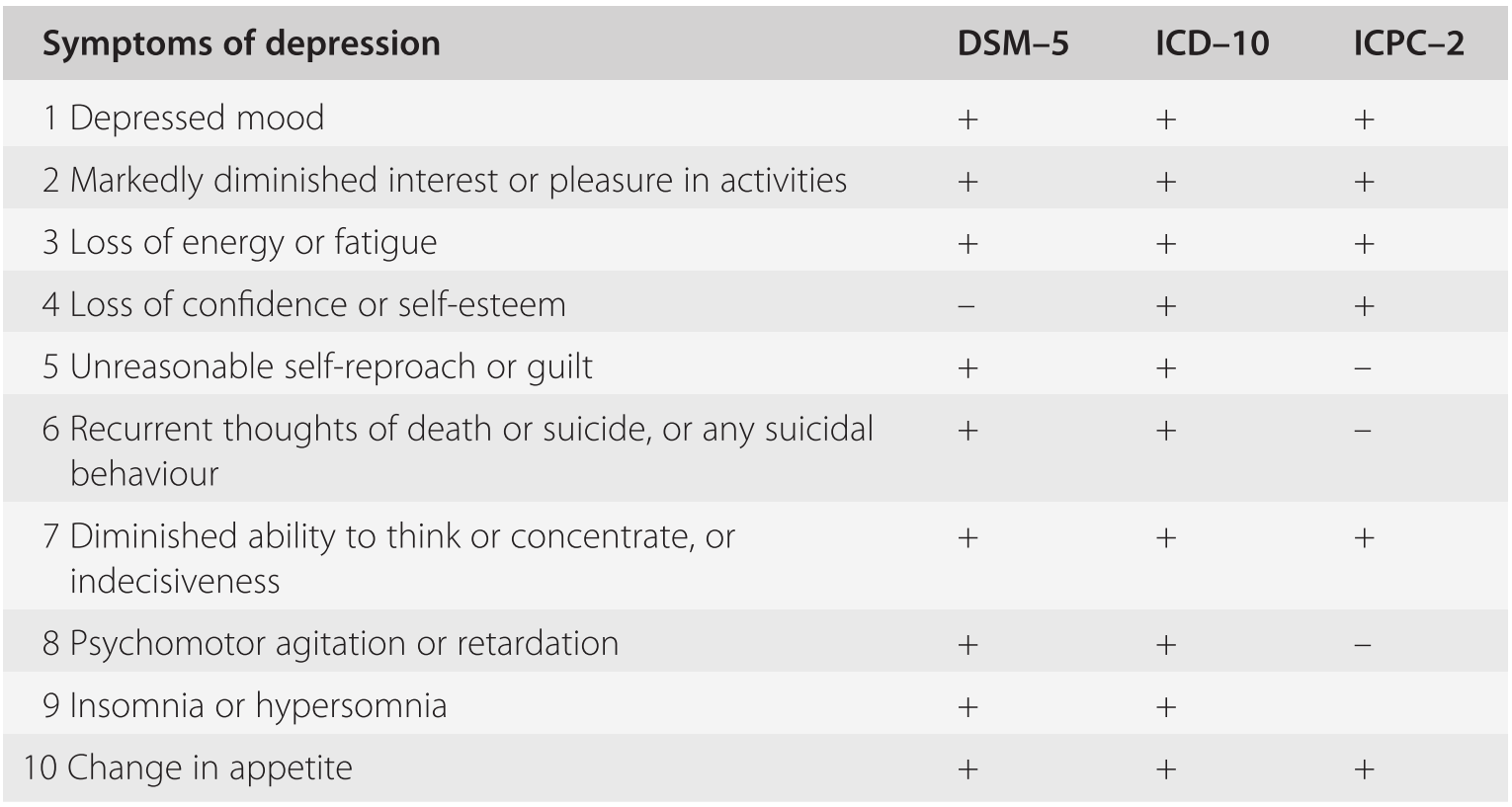
Only dispensed and administered in healthcare settings. Healthcare settings must be certified in the program and ensure that SPRAVATO ® is: Important requirements of the SPRAVATO ® REMS include the following: SPRAVATO ® Risk Evaluation and Mitigation Strategy (REMS): SPRAVATO ® is available only through a restricted program called the SPRAVATO ® REMS because of the risks of serious adverse outcomes from sedation, dissociation, and abuse and misuse. Individuals with a history of drug abuse or dependence are at greater risk therefore, use careful consideration prior to treatment of individuals with a history of substance use disorder and monitor for signs of abuse or dependence. Assess each patient’s risk for abuse or misuse prior to prescribing and monitor all patients for the development of these behaviors or conditions, including drug-seeking behavior, while on therapy.

Given its potential to induce dissociative effects, carefully assess patients with psychosis before administering SPRAVATO ® treatment should be initiated only if the benefit outweighs the risk.īecause of the risks of dissociation, patients must be monitored by a healthcare provider for at least 2 hours at each treatment session, followed by an assessment to determine when the patient is considered clinically stable and ready to leave the healthcare setting.Ībuse and Misuse: SPRAVATO ® contains esketamine, a Schedule III controlled substance (CIII), and may be subject to abuse and diversion. Sedation: In clinical trials, 48% to 61% of SPRAVATO ®-treated patients developed sedation and 0.3% to 0.4% of SPRAVATO ®-treated patients experienced loss of consciousness.īecause of the possibility of delayed or prolonged sedation, patients must be monitored by a healthcare provider for at least 2 hours at each treatment session, followed by an assessment to determine when the patient is considered clinically stable and ready to leave the healthcare setting.Ĭlosely monitor for sedation with concomitant use of SPRAVATO ® with CNS depressants (e.g., benzodiazepines, opioids, alcohol).ĭissociation: The most common psychological effects of SPRAVATO ® were dissociative or perceptual changes (including distortion of time, space and illusions), derealization and depersonalization (61% to 84% of SPRAVATO ®-treated patients developed dissociative or perceptual changes). Hypersensitivity to esketamine, ketamine, or any of the excipients. SPRAVATO ® is contraindicated in patients with:Īneurysmal vascular disease (including thoracic and abdominal aorta, intracranial and peripheral arterial vessels) or arteriovenous malformation. SPRAVATO ® is not approved for use in pediatric patients (5.5). Closely monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors.
#ICD 10 CODE FOR DEPRESSION FULL#
See full prescribing information for complete boxed warning

WARNING: SEDATION, DISSOCIATION ABUSE AND MISUSE and SUICIDAL THOUGHTS AND BEHAVIORS
